23 Feb 2023
HKU biologists and collaborators reveal a gut-brain pathway that regulates sodium appetite
A new study conducted by HKU biologists and collaborators sheds light on sodium regulation mechanisms.
A research team led by Professor Billy KC CHOW from the School of Biological Sciences at the University of Hong Kong (HKU), in collaboration with Professor Li ZHANG, Guangdong-Hong Kong-Macau Institute of CNS Regeneration, Jinan University, has uncovered a new mechanism of the Secretin (SCT) in regulating water and sodium homeostasis, which demonstrates a gut-brain pathway that initiates sodium appetite and illustrates the function of colon released SCT. The study has recently been published in a top journal– Science Advances.
Understanding sodium homeostasis in the body
Water and salt homeostasis is one of the three fundamental homeostasis for mammals to maintain body metabolism. The disorder of water and sodium homeostasis will lead to hypernatremia and hyponatremia, leading to serious organ dysfunction, including muscles and the brain. Thus, sophisticated neural and endocrine pathways are formed to regulate water and sodium balance during evolution.
Water and sodium homeostasis contain reabsorption, water consumption, and sodium appetite. The kidney controls peripheral water and sodium reabsorption mainly by the Renin-angiotensin-aldosterone system (RAAS), while water and sodium intake is tightly controlled in the brain. Circumventricular organs play vital roles in modulating water consumption, while the nucleus of the solitary tract (NTS) regulates sodium appetite.
Compared with plenty of studies that have unravelled water intake regulation in the brain, only a few studies focus on neuron pathways that regulate sodium appetite. In the brain, HSD2-positive neurons in NTS and AT1a-positive neurons in the subfornical organ (SFO) initiate sodium intake separately, while the intestine and colon take responsibility for sodium uptake. Thus, understanding gut-brain cooperation will help in better constructing complete neural pathways.
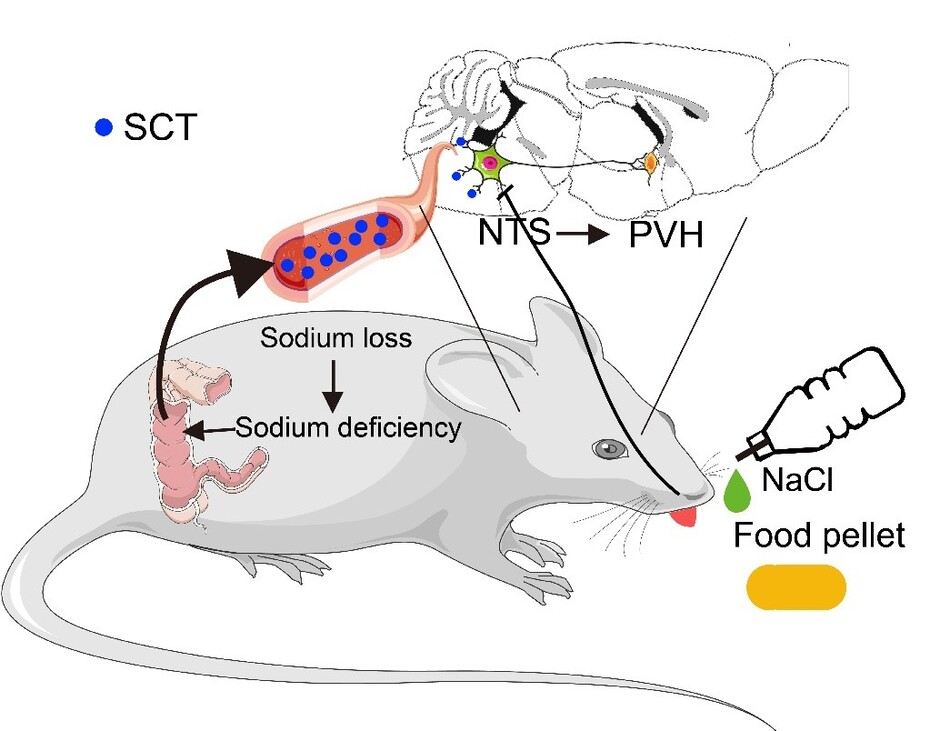
Through constant effort, our research team identified and confirmed a novel pathway that colon-released SCT activates NTS SCTR+ neurons, which project to the paraventricular nucleus of the hypothalamus (PVH) that takes responsibility for SCT-derived sodium metabolism. This study helps to better understand the peripheral-central cooperation that modulates sodium intake.
Research Background
SCT, a short peptide with only 27 amino acids, is the first identified hormone founded by William Bayliss and Ernest Starling in 1902, which regulates a wide range of systems in mammals, including gastrointestinal, cardiovascular, and central nervous systems. The SCT function is tightly connected to the SCT receptor (SCTR). As one of a class B GPCR, the signalling pathway of SCTR is closely connected with cyclic adenosine monophosphate (cAMP) and inositol triphosphate (IP3). The result from the human protein atlas shows SCTR has a high expression level in the respiratory system, GI tract, pancreas, and kidney. Some brain regions, such as NTS and the cerebral cortex, were also found SCTR expression.
When acid chyme enters the duodenum, SCT is released to the bloodstream or intestinal lumen and detected by the pancreas SCTR. The pancreas then secretes pancreatic fluid full of bicarbonate, which neutralizes the acid from the stomach and protects the intestine. In the cardiovascular system, SCT and SCTR can regulate blood pressure, cardiac blood flow, and plasma NO concentration. In the central nervous system, SCT mediates motor learning behaviour, initiates water intake, and inhibits food intake.
Key findings
Here, we revealed a gut-brain pathway in which a gastrointestinal hormone, secretin released from colon endocrine cells under body sodium deficiency condition, is indispensable for initiating salt appetite. Peripheral SCT penetrates the blood-brain barrier and enters cerebrospinal fluid. As the neural substrate, SCT activates specific receptors in the NTS, which further project to the downstream paraventricular nucleus of the hypothalamus (PVH), resulting in enhanced sodium intake. These results demonstrated a previously unrecognized gut-brain pathway for the timely regulation of sodium homeostasis.
Our results also have potential values in clinical fields, as ileostomy patients showed chronic salt and water disorders, together with low urinary sodium excretion, all of which are similar to the mice under sodium deficiency conditions. Such clinical observations can be replicated in animal models, as previous studies indicated SCT expression increased within 24 hours after colectomy and decreased significantly on day 14 in rats. Although no direct evidence has been provided for the role of colon-SCT in sodium homeostasis in human patients, these studies did imply its possible involvement. Further work can be performed to explore the possibility of targeting SCT-SCTR for correcting sodium imbalance in colectomy patients.
The journal paper can be accessed here.
About the research team

The research was conducted by the team led by Professor Billy KC Chow from HKU School of Biological Sciences. Dr Yuchu LIU from Professor Chow’s team is the first author of the paper. Professor Li Zhang is the co-corresponding author, and his team make a great effort in this study.
The research has been supported by three grants from the Research Grants Council of Hong Kong and the National Natural Science Foundation of China. This study was supported by the Research Grants Council of Hong Kong GRF HKU17113120, HKU17127718, and the National Natural Science Foundation of China 32070955.
About Professor Billy Chow
Professor Billy K. C. Chow is the Chair of Endocrinology at the School of Biological Sciences, HKU. His expertise lies in GPCR and ligand interactions, eventually leading to deciphering the physiologic actions of GPCRs and identifying novel GPCR agonists/antagonists as therapeutic solutions. He is the founder of an HKU spin-off company PhrmaSec Ltd, which aims to translate basic research into societal impact by targeting GPCRs.